- Blog
- About
- Contact
- Age of empires 3 mac
- Android 6-0 marshmallow zip file
- Rhonda byrne the secret audiobook free download
- Native instruments maschine mikro mk2 white
- New marathi song
- Galaxy note 4 imei repair tool
- Hulk loki pis art of war comicvine
- Screen image capture mac
- Mods for universal minecraft editor pastebin
- Download dota 2 offline kaskus
- Muvizu app hang
- Serum osmolality
- Uleads photoimpact
- Saints row 2 coop
- Ram leela movie online watch
- Pilsner beer recipe

This is due to pseudohyponatraemia, an apparent low measured sodium concentration (see The calculated osmolarity will differ from the measured osmolality in specimens with high lipid (lipemic) or protein (e.g.
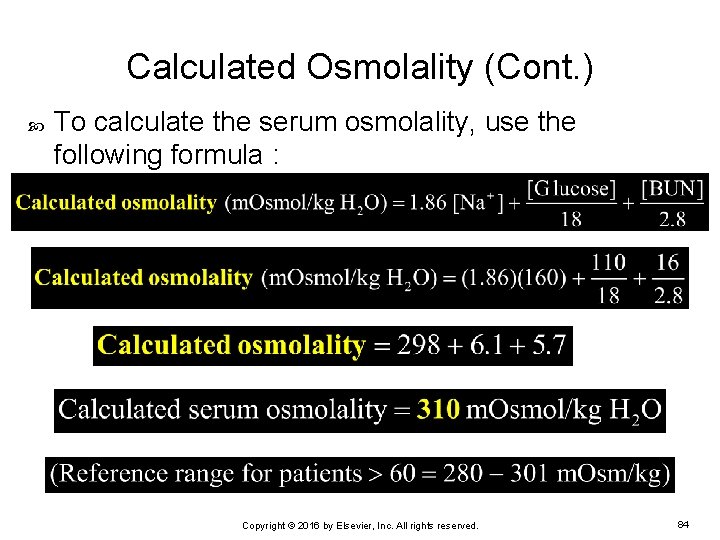
An increased osmolar gap may be due to the presence of mannitol, ethanol, methanol, ethylene glycol, or other substance. The normal range for the osmolar gap is 0 - 15. : The osmolar gap is the difference between the measured and calculated serum osmolalities. ):Īn alternative formula is: (Na x 2) + Urea + Glucose. The following formula is used in the laboratory computer calculation (ref: Weisberg, Clin Chem (1975) 21/8 1182. This can be a useful pointer to the presence of ethanol, mannitol or other solutes in the plasma. When sodium, urea and glucose results are available, these can be used to calculate an estimated osmolality. When osmolality is requested on a sample, this will be measured by an osmometer. They may also order additional tests or exams.DISCLAIMER: This link was displayed at 22:45:26 and expires on if printed.Īlso known as : ,ĬC6548 Fluid Deprivation Laboratory Request FormĠ.5 mL Paediatric Micro-PST Blood (Preferred) Your doctor will use the results of your test to help develop a diagnosis. Some of these causes are less serious than others.

Physicians may also use this test in consideration with the measured amounts of blood urea nitrogen, glucose, and sodium in your serum. The serum test is used mainly to evaluate hyponatremia, a below normal level of sodium in the bloodstream. The blood osmolality test is also known as a serum osmolality test. This helps your body get rid of excess water, raising the osmolality of your blood.

For example, you may need to urinate frequently if your blood osmolality is too low. When your body is functioning properly, it makes specific adjustments to maintain an appropriate osmolality. Very salty water has higher osmolality than water with just a hint of salt. The greater the concentration of the substance dissolved, the higher the osmolality. Osmolality is a measure of how much one substance has dissolved in another substance.
